The Case That Started the Debate
He was not improving.
At least, that is how the chart described it.
A 52-year-old patient with recurrent depression and chronic anxiety had already undergone multiple medication trials. His current regimen included an SSRI, a low-dose atypical antipsychotic for augmentation, a benzodiazepine for sleep, and later a mood stabilizer added for “partial response.”
Each medication had a clear rationale.
Each had a defensible mechanism of action.
Yet his functioning declined.
Cognitive slowing increased.
Fatigue deepened.
Anxiety paradoxically intensified.
Eventually, the label of treatment resistance was applied.
But an uncomfortable question lingered:
Was the illness resistant, or was the pharmacology colliding?
This is the modern psychiatric dilemma.
In an era where guideline adherence, symptom scales, and rapid prescribing pathways dominate care, the relationship between mechanism of action and polypharmacy is becoming one of the most controversial and clinically consequential debates in psychiatry.
Mechanism of Action in Psychiatry: The Biological Language We Sometimes Ignore
Every psychiatric medication exerts its effect through a specific mechanism of action, whether through receptor modulation, transporter inhibition, ion channel effects, or neuroplastic signaling pathways.
SSRIs inhibit serotonin reuptake transporters.
Antipsychotics modulate dopamine D2 receptors and often multiple serotonergic pathways.
Mood stabilizers influence intracellular signaling cascades and neuronal excitability.
Benzodiazepines enhance GABAergic inhibitory tone.
On paper, combining mechanisms appears logical.
In practice, however, mechanisms do not operate in isolation.
They interact.
They compete.
They amplify or blunt each other.
And in polypharmacy, the cumulative biological signal can become unpredictable.
According to the American Psychiatric Association, polypharmacy rates in psychiatric populations have increased substantially over the past two decades, particularly in mood and psychotic disorders. Observational data suggest that many patients receive three or more psychotropic medications concurrently, often without clear mechanistic integration.
The question is not simply whether medications are evidence-based.
It is whether their combined mechanisms of action form a coherent biological strategy.
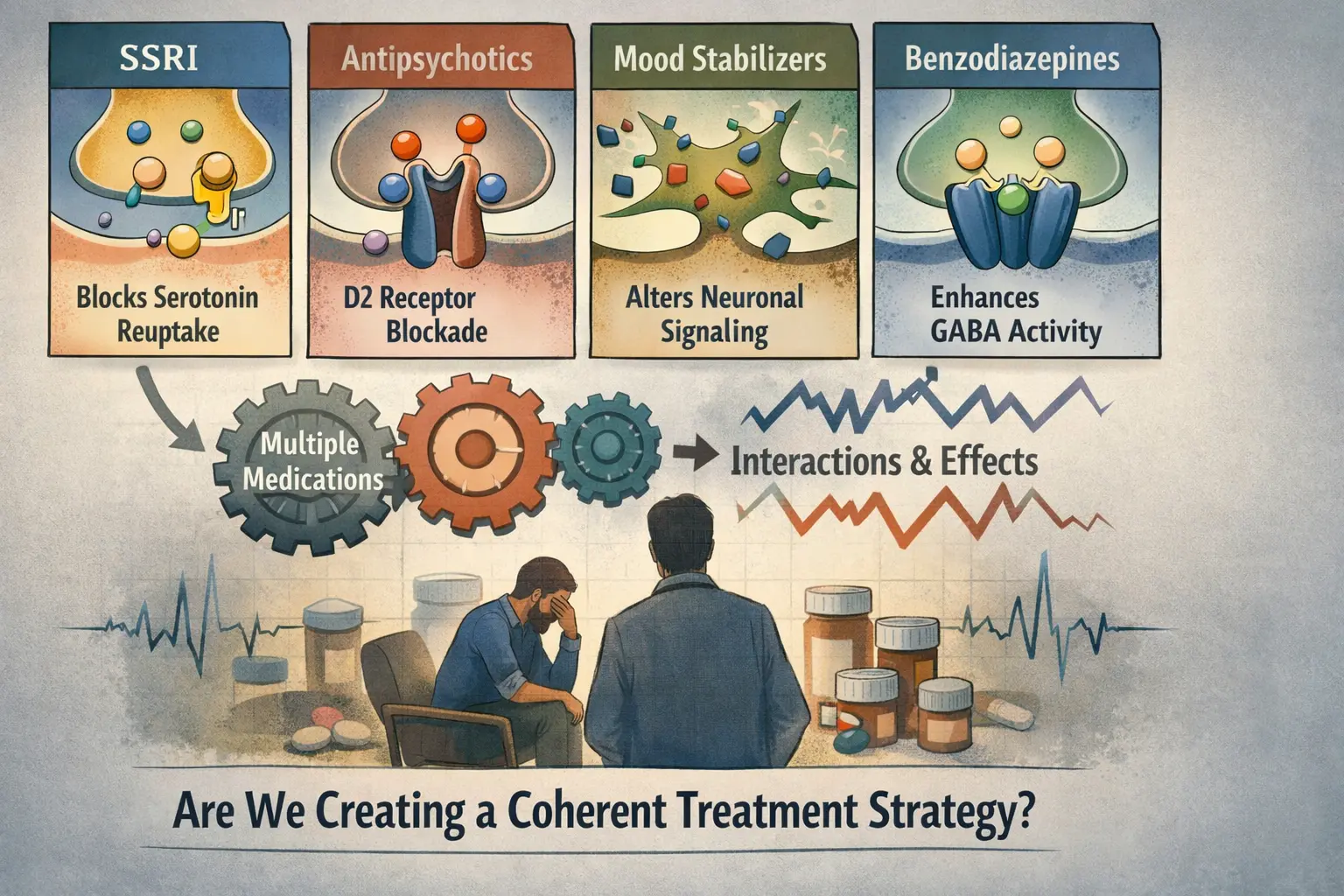
Mechanism of Action and Polypharmacy: When Pharmacodynamics Begin to Overlap
One of the most underrecognized drivers of complex psychiatric outcomes is pharmacodynamic redundancy.
This occurs when multiple medications influence similar receptor systems or neurochemical pathways without producing additive therapeutic benefit.
Examples include:
Dual serotonergic agents increasing risk of serotonin toxicity without improving remission rates
Antipsychotic stacking leading to excessive D2 blockade and cognitive dulling
Sedative layering causing functional impairment mistaken for depressive relapse
These patterns illustrate a critical principle:
More mechanisms do not necessarily translate into more therapeutic signal.
In fact, they may dilute it.
CANMAT guidelines emphasize structured sequencing of pharmacologic strategies in mood disorders, yet real-world prescribing often deviates due to symptom urgency, partial response anxiety, or clinician uncertainty.
The result is a growing phenomenon:
Polypharmacy driven not by disease complexity alone, but by mechanistic ambiguity.
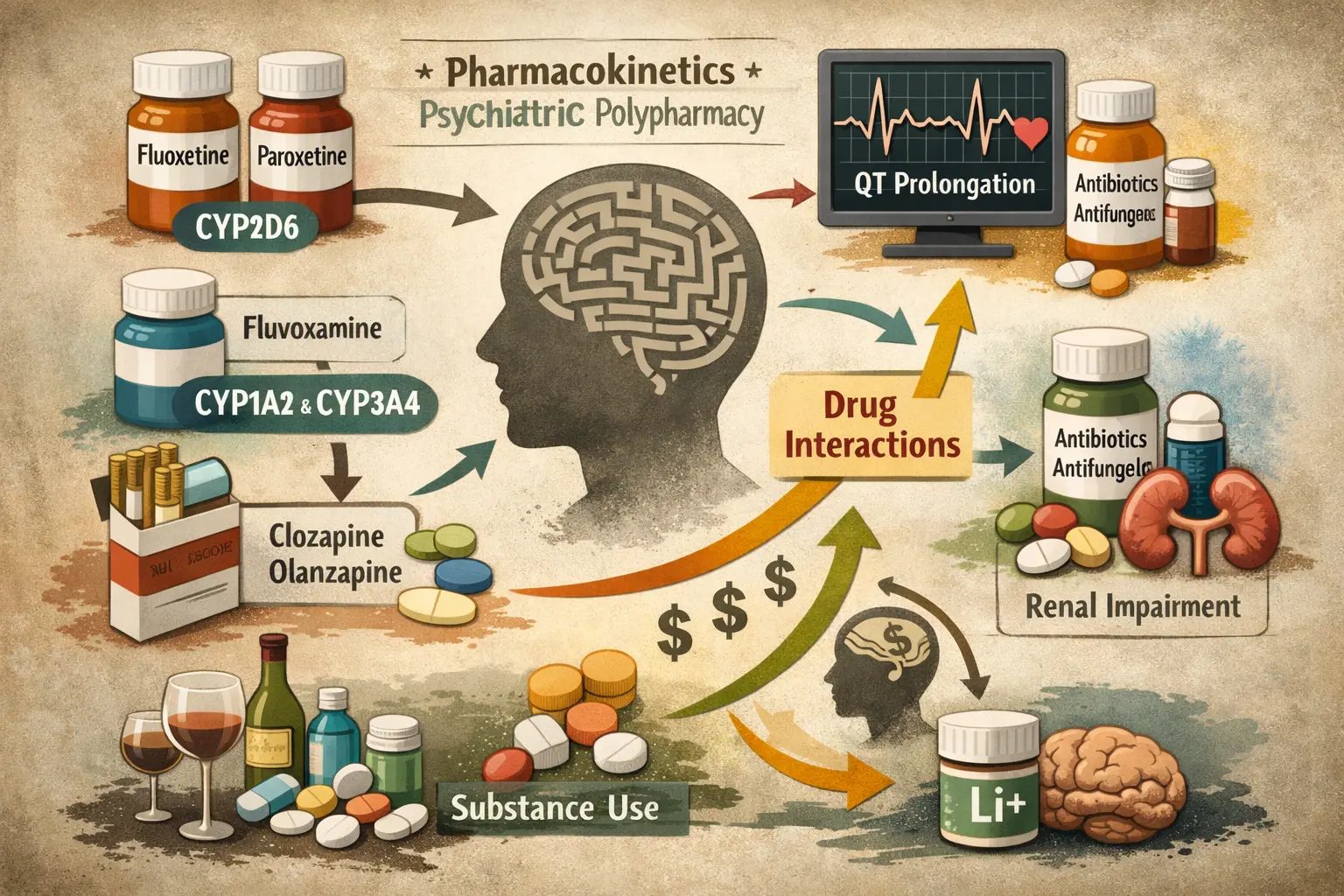
Pharmacokinetics: The Invisible Variable in Psychiatric Polypharmacy
While pharmacodynamics explains receptor-level interactions, pharmacokinetics determines how drugs are absorbed, metabolized, and eliminated.
In psychiatric practice, this often means navigating the intricate world of CYP450 metabolism.
Fluoxetine and paroxetine inhibit CYP2D6.
Fluvoxamine inhibits CYP1A2 and CYP3A4.
Smoking induces CYP1A2 metabolism, lowering levels of medications such as clozapine and olanzapine.
Macrolide antibiotics or antifungals may increase QT-prolonging antipsychotic concentrations.
In polypharmacy, these effects can cascade.
A new medication may unintentionally elevate another drug’s plasma level.
A change in substance use may destabilize previously stable regimens.
Renal impairment may amplify lithium exposure despite unchanged dosing.
The American Society of Addiction Medicine highlights similar interaction risks in dual-diagnosis populations, where psychotropics intersect with substances such as alcohol, stimulants, or opioids.
These pharmacokinetic dynamics transform medication regimens into evolving biological systems.
And often, clinicians are reacting to downstream consequences rather than anticipating upstream mechanisms.
Mechanism of Action vs Symptom-Based Prescribing: A Modern Controversy
Another controversial dimension of psychiatric polypharmacy lies in the tension between mechanism-driven prescribing and symptom-driven prescribing.
Symptom escalation frequently leads to medication addition.
Sleep worsens → sedative added.
Anxiety increases → anxiolytic added.
Motivation declines → stimulant considered.
Each intervention addresses a surface phenomenon.
Yet the underlying biological system may remain unchanged or further destabilized.
Pioneering psychopharmacology researchers have long emphasized that treatment-resistant syndromes often reflect heterogeneous biological pathways rather than simple neurotransmitter deficits.
In other words:
The mechanism of action matters more than the medication count.
This raises an uncomfortable clinical question:
Are we sometimes treating medication side effects with more medications?
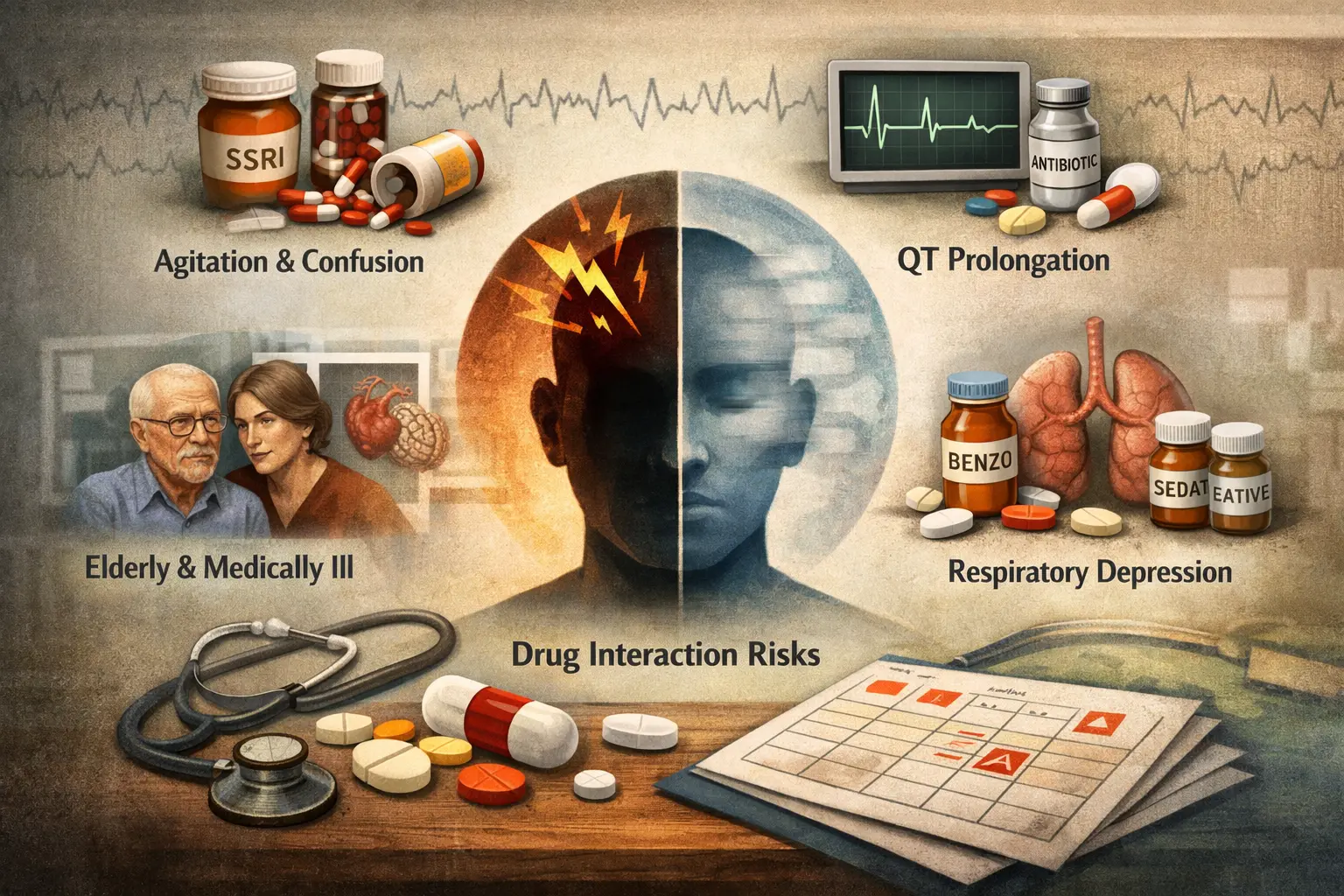
Drug-Drug Interaction Syndromes: When Polypharmacy Creates New Illness Patterns
One of the most compelling and least discussed realities of psychiatric practice is the emergence of interaction syndromes.
These are clinical presentations driven not by primary psychiatric illness but by combined pharmacologic effects.
Examples include:
Mixed agitation states caused by serotonergic overstimulation
Cognitive slowing from cumulative anticholinergic burden
QT prolongation risk from combined antipsychotic and antibiotic therapy
Respiratory suppression risk from benzodiazepine and sedative-hypnotic combinations
In geriatric populations, polypharmacy magnifies vulnerability due to reduced hepatic metabolism and renal clearance.
In medically ill patients, cardiovascular or neurological consequences may dominate psychiatric symptomatology.
Despite guideline awareness, real-world detection of such interaction patterns remains inconsistent.
This is where structured drug-drug interaction reasoning frameworks become critical.
The Sequencing Problem: When Timing Alters Mechanism
Polypharmacy is not merely about how many medications are prescribed.
It is about when they are introduced.
Neuroadaptation processes, receptor upregulation, downstream signaling changes, and synaptic plasticity shifts occur over weeks to months.
Premature augmentation may interrupt biological stabilization.
Rapid switching may trigger withdrawal phenomena mistaken for relapse.
Delayed initiation of definitive treatments such as clozapine or ECT may prolong illness burden.
APA guidance repeatedly emphasizes the necessity of adequate trial duration and dose optimization before medication layering.
Yet in busy clinical settings, therapeutic decisiveness can be overshadowed by pressure for rapid symptom resolution.
This creates a paradox:
Polypharmacy may increase, not decrease uncertainty.
Mechanism of Action Integration: A Clinical Skill, Not Just Pharmacology Knowledge
The solution is not to avoid combination treatment altogether.
There are many scenarios where polypharmacy is life-saving.
Bipolar disorder stabilization may require a mood stabilizer plus an antipsychotic.
Treatment-resistant depression may benefit from carefully chosen augmentation strategies.
Severe anxiety disorders may require temporary multi-mechanism approaches.
The critical factor is mechanistic coherence.
Each medication should contribute a distinct biological signal.
Each interaction risk should be anticipated.
Each pharmacokinetic variable should be monitored.
This is where emerging digital tools including psychiatric clinical co-pilot platforms such as On-Demand Psychiatry are beginning to reshape decision support.
By integrating receptor-level pharmacology, CYP metabolism modeling, QT risk detection, and guideline sequencing logic, such systems can help clinicians visualize the mechanistic architecture of a regimen before consequences emerge.
In complex cases, this may represent the difference between targeted combination therapy and biologically chaotic polypharmacy.
The Unresolved Debate: Is Polypharmacy a Marker of Severity or Systemic Blind Spots?
The controversy remains.
Some argue rising polypharmacy reflects increasing illness complexity.
Others contend it reflects fragmented care pathways and limited time for deep mechanistic reasoning.
Both may be true.
What is clear is that psychiatric prescribing is evolving.
Clinicians are managing more comorbidity.
Patients are living longer with chronic psychiatric illness.
Substance use patterns are shifting.
Medical comorbidities are increasing.
In this environment, understanding the mechanism of action as an integrated system variable becomes essential.
Polypharmacy is not inherently harmful.
But polypharmacy without biological strategy can be.
FAQ: Mechanism of Action and Polypharmacy in Practice
Is psychiatric polypharmacy always inappropriate?
No. Many severe psychiatric conditions require multi-mechanism treatment. The key is ensuring each medication contributes a distinct therapeutic pathway.
How do drug-drug interactions influence treatment outcomes?
Interactions can alter drug levels, amplify side effects, or reduce efficacy. Pharmacokinetic monitoring and mechanistic awareness are essential.
Can clinical decision-support tools help manage complex regimens?
Yes. Platforms such as On-Demand Psychiatry are designed to assist clinicians by identifying pharmacodynamic overlap, CYP metabolism risks, QT interactions, and safer sequencing strategies.
Why is mechanism of action more important than medication count?
Because psychiatric outcomes depend on biological signal clarity. Multiple drugs targeting similar pathways may create noise rather than therapeutic synergy.
The Resolution: Precision Lies Not in Prescribing More But in Prescribing Smarter
Psychiatry is entering a new era.
One where medication selection is no longer defined solely by diagnosis.
One where symptom scales alone cannot guide decision-making.
One where timing, metabolism, receptor dynamics, and interaction patterns must be synthesized in real time.
The future of psychiatric treatment will not be measured by how many medications we can combine.
It will be measured by how precisely we can align the mechanism of action with individual neurobiology.
Polypharmacy may remain necessary.
But mechanistic clarity must lead it.
Because in the end, the most powerful intervention in psychiatry is not simply choosing the right drug.
It is important to understand how every drug in the system interacts with the brain at the same time.
And when that language becomes coherent, outcomes begin to change.

This article is intended for licensed healthcare professionals. It does not provide medical advice, diagnose conditions, or substitute for clinical judgment. All clinical decisions must be made by a qualified clinician familiar with the individual patient. For emergencies, call 911. For mental health crisis support in the US, call or text 988.
.webp)


.webp)